|
Theory |  |
Exercises |  |
 |
Lection 0: Electric Charge
In this lecture, the electric charge is introduced.
Goal: The goal of this lecture is to have a basic understanding of atoms and its structure. Emphasis is layed on the definition of electric charge and the charge of an atom as this is the essence of electric current.
|
To understand electric charge, it is important to understand know what atoms are and the structure of atoms. The ancient greeks invented the term atom, it means invisible. They thought, that everything is made up of small particles. But the particles are so small, that they must be invisible. |
|||||
|
With this assumption, they were almost correct.
Atoms are the particles all matter is built from.
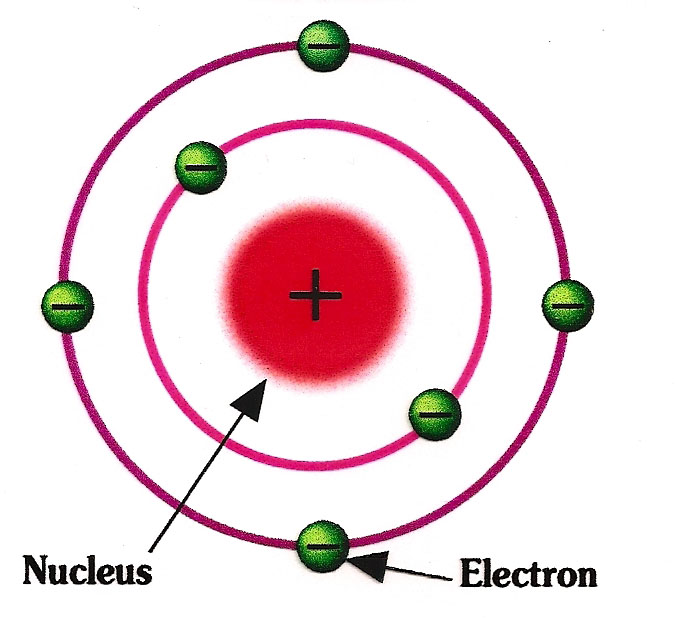
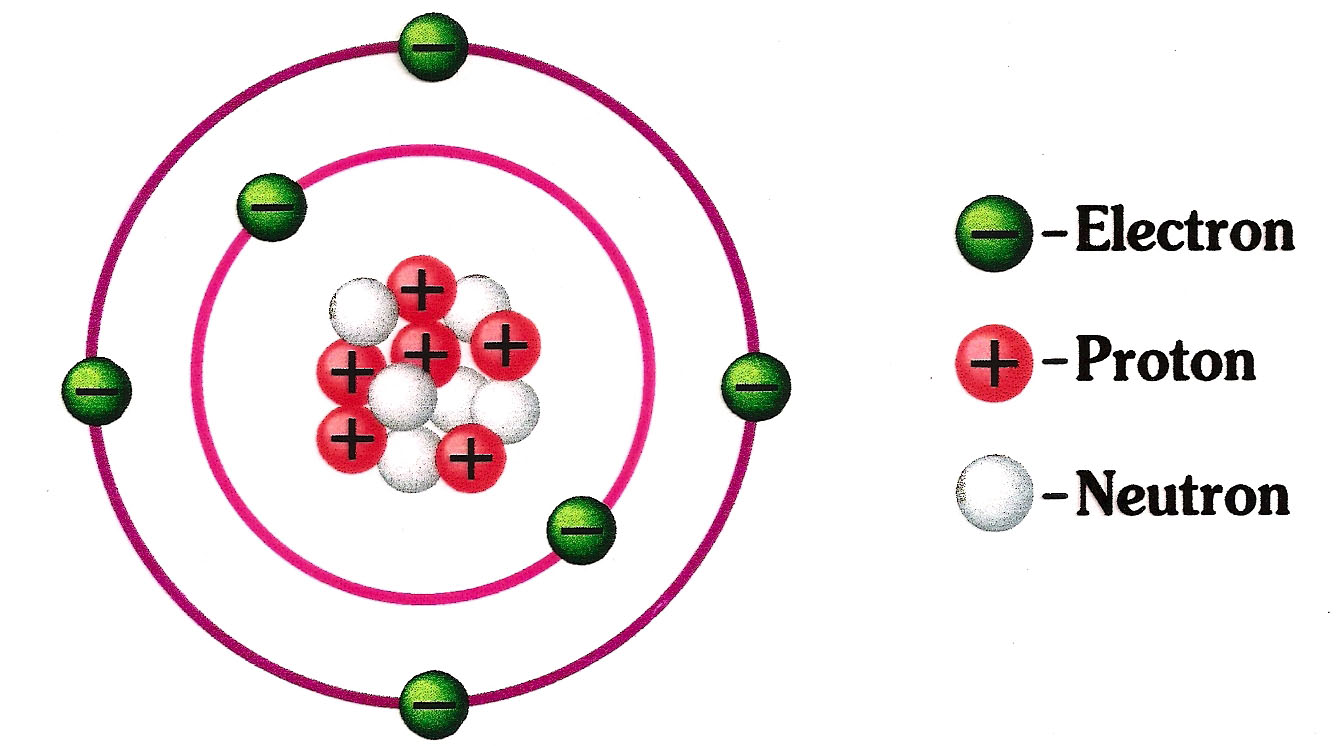
The periodic table gives an overview over all existing atoms. But ithe atoms can be further divided into even smaller subatomic parts. Subatomic means part of an atom. In 1911, a British physicist called Ernest Rutherford formulated the theory of the hull model about how atoms look like. The hull model says that every atom is made up of a centre and an outer hull. The centre is often called by its greek name nucleus. In the nucleus (the centre or core of the atom) there are two types of subatomic particles. The protons and the neutrons. In the outer hull there is another type of subatomic particle, the electron. In the second picture on the left, the red balls represent the protons and the white balls the neutrons. These two parts make up the nucleus of of the atom. The green balls are the electrons and are in the outer hull of the atom. The protons and neutrons are about equally heavy and together, these make up for almost all of the mass of the atom. In fact, about 99.94 % of the total mass of the atom is only coming from the protons and neutrons. The electrons are very small and light in comparison and make up only about 0.06 % of the mass of the atom. One can imagine the atom as ball with a very thin hull but a very heavy small center. Now, the different subatomic particles have different charges. The neutron, which is in the nucleus of the atom, has no charge, we call this particle neutral. The name neutron actually means neutral in ancient greek. The proton, which is also in the nucleus, has a positive charge. The electron, which is in the outer hull of the atom, has a negative charge. |
||||
|
A positive particle always attracts a negative particle, but repulses another positive particle. And a negative particle attracts a positive particle but repulses another negative particle. Or to say it quicker, same charges repulse each other while opposite charges attract each other. This is similar to magnets. A magnet has a positive and a negative side. The positive side always attracts a negative side while it repulses another positive side. Vice versa for negative sides. The rule is that same sides repulse each other while opposite sides attract each other. Every proton hasthe power to attract one electron. But no more. If a proton is attracting one electron, it is not strong enough to attract another electron. This is because the strength of the positive charge of the proton is ecactly the same as the strength of the negative charge of the electron. So if a proton is attracting one electron, one can say that the positive charge of the proton and the negative charge of the electron are neutralising each other. Other electrons or protons will not see any charge of this pair. The pair is now balanced. As mentioned before, every electron has one negative charge and every proton has one positive charge. If we look at the carbon atom presented in the picture above, we see that it has six electrons and six protons. So the number of negative charge is equal to the number of positive charge. If the total positive charge is exactly the same as the negative charge, then the atom is balanced because every proton attracts exactly one electron. From the outside, the atom has no charge, it is neutral. But if the atom looses an electron for whatever reasons, the total charge of the atom would be +1 as it has one proton more that electrons. Or one could say, one proton has no electron to attract and therefore its charge is not neutralized by a negative charge. The opposite happens is a neutral atom (same number of protons as electrons) gains an electron. Then it has a negative charge of -1 because one electron in the atom has no proton to be attracted to and its negative charge is not neutralized. By the way, atoms that are not balanced are called ions. That means, if an atom has more electrons than protons it has a negative charge and is over all a negative ion. If it has more protons than electrons, it has a positive charge ans is over all a positive ion. |
|||||
Conclusion and short repetitionLet's briefly list all the important information from this chapter in a short list.
|